The rise of mRNA-based vaccines has revolutionized global health but poses challenges for purification platforms. Traditional methods struggle with hazardous solvents, high salt concentrations, and scalability. Achieving high purity is essential, especially for mucosal mRNA vaccines, where double-stranded like impurities can affect efficacy and trigger an unwanted immune response.
This blog explores how monolithic columns transform mRNA purification, addressing these challenges and ensuring high purity and yield. Discover their unique features and impact on mRNA integrity, making them ideal for vaccine development and production.
Hybridization Affinity Chromatography: A Superior Approach
Hybridization affinity chromatography, utilizing immobilized oligo-deoxythymidilic acid (oligo dT) ligands, offers a pivotal method for mRNA capture. This technique allows specific binding of mRNA with a poly-A tail, ensuring high purity and yield without denaturing conditions. It effectively separates impurities and mRNA fragments lacking a poly-A tail, preserving mRNA integrity.
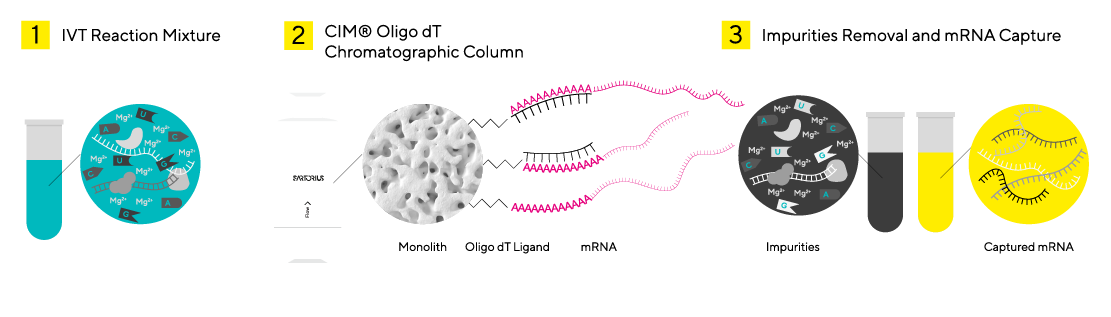
Innovative Convective Flow for Enhanced Purification
Monolithic columns with interconnected channels enable mass transfer predominantly via convective flow, overcoming limitations of traditional matrices. Unlike conventional methods that require residence times over 60 seconds, these columns achieve residence times under 10 seconds, allowing operation at higher flow rates with minimal reduction in dynamic binding capacity. This consistency and reliability make them ideal for mRNA purification at room temperature, effectively addressing the instability of mRNA in the IVT matrix.
Efficiency and Stability in mRNA Purification
Monolith-based advanced chromatography techniques deliver over 95% yield and high quality of eluted mRNA, effectively removing impurities like T7 RNA polymerase and over 99.5% of double stranded molecules. This high purity is crucial for enhancing mRNA efficacy and reducing inflammation, especially in mucosal applications.
The robust stability of these matrices, even after prolonged treatment with 0.5 M NaOH and long-term storage in 20% ethanol, makes them ideal for industrial applications, helping to reduce production costs.
Scalability for Industrial Applications
Monolithic columns offer exceptional scalability, expanding mRNA purification from 0.1 mL to at least 800 mL column volumes, and paving the way for manufacturing processes with 40 L column volumes. Their short residence times increase throughput and allow multi-cycling, reducing costs and space requirements in manufacturing facilities. This flexibility is vital for mRNA vaccine development and manufacturing, ensuring high-quality production.
Why Test Monolithic Columns?
These columns offer an effective solution for mRNA purification by combining efficiency, stability, high yield, and scalability. They operate at high flow rates with minimal dependence on residence time and mRNA size and reliably remove IVT reaction components and impurities related to double-stranded species. This makes them a top choice for producing high-quality mRNA, which is crucial for meeting the growing demand for mRNA therapeutics and vaccines while reducing production costs.
Read our whitepaper, “Simplify mRNA Purification for Faster, Scalable Production,” to gain deeper insights into optimizing your mRNA purification strategy with proven, scalable technology.
Jingjing Zhang, Chao Li, Yuheng Liu, Rui Liao, Dian He, Lifeng Xu, Tingting Chen, Qin Xiao, Mingxing Luo, Yang Chen, Yali Li, Huaxing Zhu, Joseph Rosenecker, Xiaoyan Ding, Shuchen Pei, Shan Guan. Airway applied IVT mRNA vaccine needs specific sequence design and high standard purification that removes devastating dsRNA contaminant
Dewar EA, Guterstam P, Holland D, Lindman S, Lundbäck P, Brito Dos Santos S, Wang SC, Swartz AR. Improved mRNA affinity chromatography binding capacity and throughput using an oligo-dT immobilized electrospun polymer nanofiber adsorbent
Nina Mencin, Dona Štepec, Alja Margon, Jana Vidič, Darko Dolenc, Tina Simčič, Sara Rotar, Rok Sekirnik, Aleš Štrancar, Urh Černigoj. Development and scale-up of oligo-dT monolithic chromatographic column for mRNA capture through understanding of base-pairing interactions




