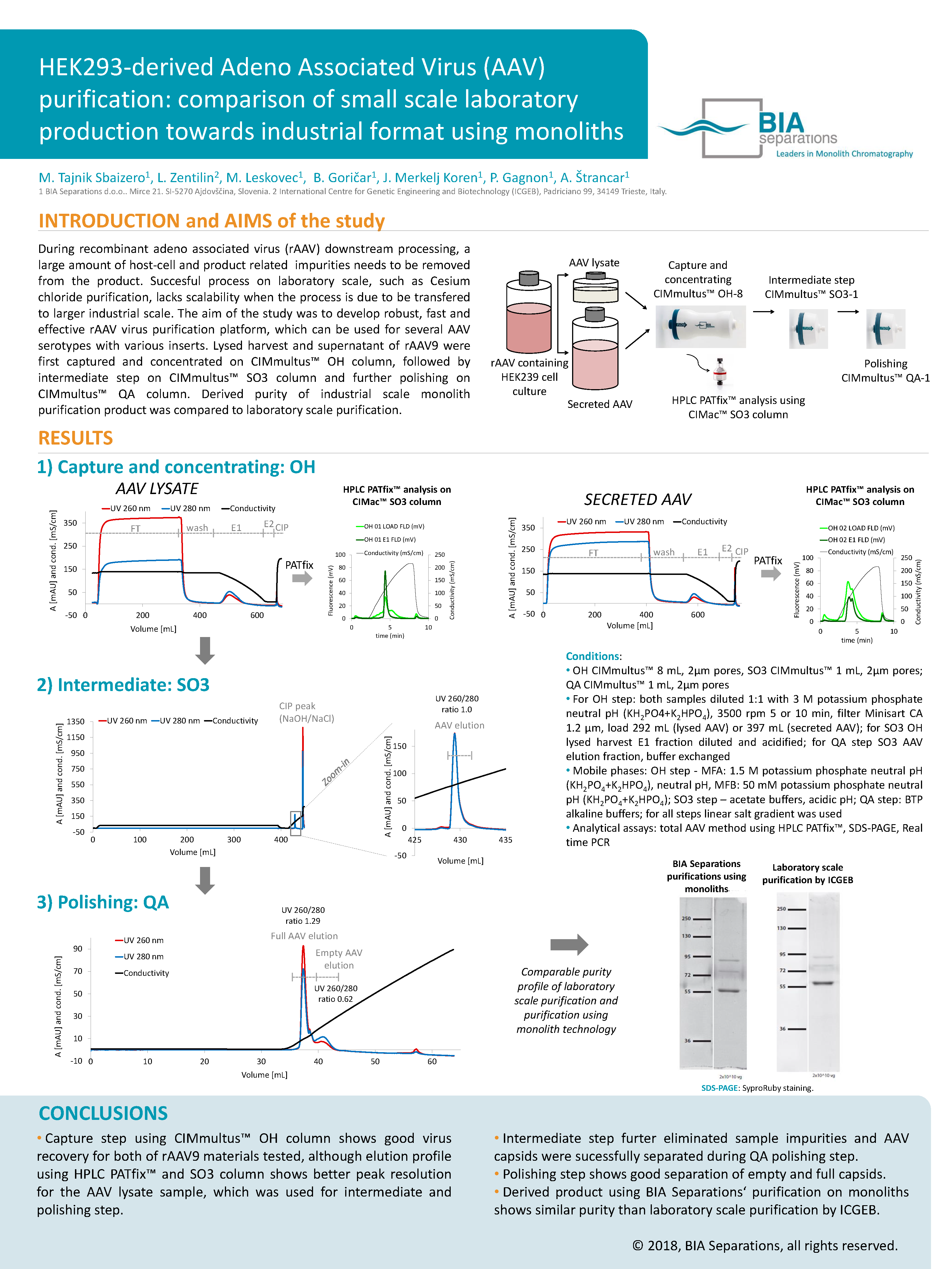
During recombinant adeno associated virus (rAAV) downstream processing, a large amount of host-cell and product related impurities needs to be removed from the product. Successful process on laboratory scale, such as Cesium chloride purification, lacks scalability when the process is due to be transferred to larger industrial scale.
The aim of the study was to develop robust, fast and effective rAAV virus purification platform, which can be used for several AAV serotypes with various inserts. Lysed harvest and supernatant of rAAV9 were first captured and concentrated on CIMmultus OH column, followed by intermediate step on CIMmultus SO3 column and further polishing on CIMmultus QA column. Derived purity of industrial scale monolith purification product was compared to laboratory scale purification.